Three years in, the supply chain is shrinking, the timeline is slipping, and patients are turning to the black market. Washington’s fix isn’t working.
This is the third installment in Riptide’s ongoing coverage of the Adderall shortage. Part One examined the structural origins of the crisis. Part Two covered the DEA’s January quota increase and its limits. This update reports on what has changed — and what has gotten worse — since then.
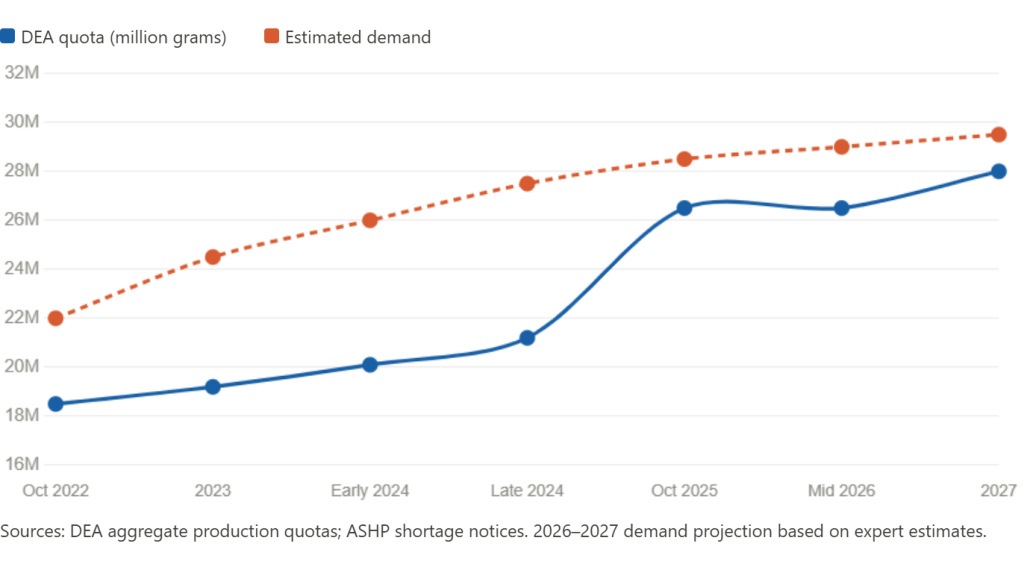
In October 2025, the Drug Enforcement Administration announced what sounded like a turning point. The agency raised its aggregate production quota for d-amphetamine from 21.2 million grams to 26.5 million grams — a 25 percent increase, the first significant adjustment in years. The announcement was treated as a signal that Washington had finally acknowledged what patients had been living with since October 2022: a shortage that had no end in sight.
That signal was premature.
As of May 2026, the American Society of Health-System Pharmacists still lists both immediate-release and extended-release amphetamine mixed salts in active shortage. The FDA’s original shortage notice, first posted in October 2022, remains live. The shortage has now entered its fourth year. And the picture that has emerged since January is not one of a system slowly catching up — it is one of a supply chain quietly contracting around a problem that regulators have not fully solved.
The Quota Increase and the Gap Behind It
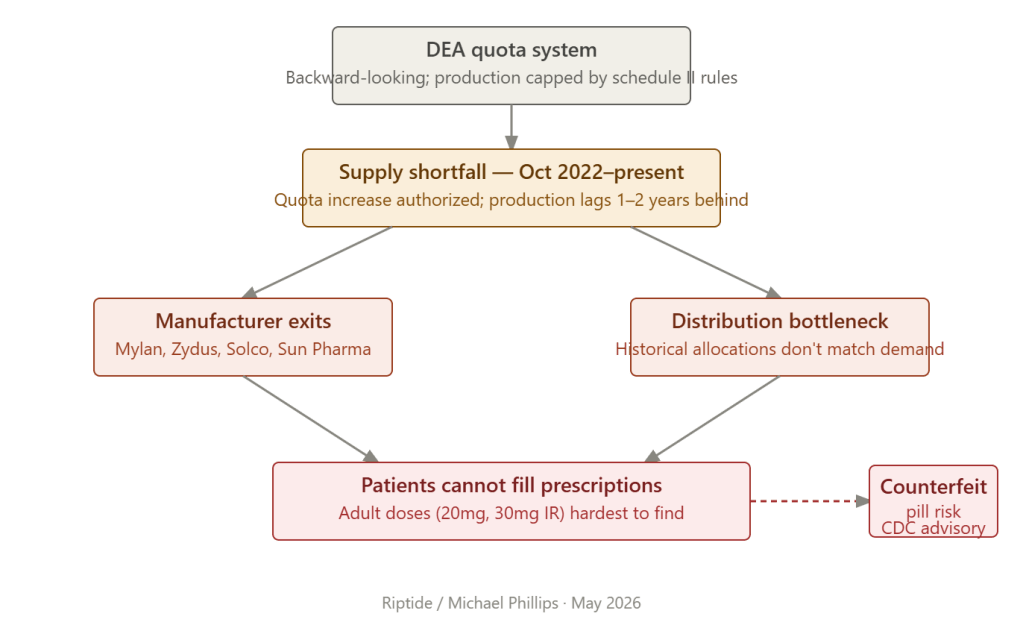
A production quota increase is not a medication increase. It is permission — permission for manufacturers to produce more, contingent on their ability to acquire the raw materials, schedule the production runs, and move finished product through a distribution system still governed by historical allocation formulas.
The gap between what the DEA authorized and what patients can actually find at a pharmacy has not closed on anything close to the timeline implied by the October announcement. Experts now estimate it could take until late 2026 or 2027 before supply consistently meets demand nationwide. That is a two-to-four-year lag between the DEA recognizing the problem and American patients reliably being able to fill their prescriptions.
Part of the delay is structural. Pharmaceutical manufacturers source the active pharmaceutical ingredients — the APIs that form the chemical basis of amphetamine medications — through global supply chains that are themselves subject to bottlenecks. A delay at a single overseas plant can cascade through multiple domestic manufacturers simultaneously. Raising the quota does nothing to accelerate that upstream supply.
Part of it is the distribution system itself. Drug distributors allocate controlled substances to pharmacies based on historical ordering patterns. A pharmacy that has seen a recent spike in ADHD patients — or one that is simply newer — may receive allocations that do not reflect its actual patient load. The quota increase flows into a distribution architecture that was not designed for the demand environment it is now navigating.
The Generic Market Is Shrinking, Not Growing
The most underreported development since January is not the pace of the quota increase. It is what has happened to the generic manufacturer base.
Mylan and Zydus have both discontinued their immediate-release amphetamine mixed salt tablets. Solco has also exited the market. Sandoz remains in shortage due to increased demand. Sun Pharma and Sandoz have discontinued their extended-release capsule lines. What remains on the ASHP’s available manufacturer list for extended-release is a short one: Shire’s branded Adderall XR, Lannett, Aytu BioPharma’s Adzenys XR ODT, and Tris Pharma’s Dynavel XR.
This is the opposite of what a functioning market response to a shortage looks like. In a normal supply-demand scenario, a persistent shortage creates an incentive for new entrants and expanded production from existing players. What has happened instead in the generic amphetamine market is consolidation through exit. Companies are leaving, not arriving.
The reasons are not hard to identify. Manufacturing Schedule II controlled substances requires DEA registration, specialized facilities, tight regulatory compliance, and exposure to quota ceilings that can limit return on investment before a production line ever reaches full capacity. The regulatory architecture that was meant to prevent diversion is also, structurally, a barrier to the market response that patients need.
The doses hit hardest by this contraction are adult doses. The 20 mg and 30 mg immediate-release tablets — the most commonly prescribed strengths for adults — remain the hardest to locate. For extended-release, the 15 mg, 25 mg, and 30 mg capsules have been the most consistently in short supply. The patients most systematically unable to fill their prescriptions are working-age adults.
The Harm the Shortage Is Producing
When the system fails to reliably provide medication, patients do not simply go without. They adapt — and some of those adaptations carry their own serious risks.
The CDC issued health advisories in 2024 and 2025 flagging a pattern that has emerged directly from the shortage: increased counterfeit pill circulation among patients who cannot obtain legitimate prescriptions. Counterfeit amphetamine products are frequently pressed with fentanyl or other adulterants. The patient who has exhausted 25 pharmacies, as one documented case involved a 22-year-old college student, faces a choice between going unmedicated and seeking supply through channels the government cannot regulate.
This is the harm calculus that the DEA’s quota-setting framework does not appear to weigh. The regulatory system was designed around the risk of diversion — the concern that legitimate prescription channels would be exploited to produce a recreational supply. What it did not adequately model is the risk created by the shortage itself: that patients cut off from the legitimate supply chain would turn to an unregulated one.
The CDC’s advisory noted particular concern about injury risk and overdose vulnerability among patients experiencing disrupted stimulant access. For patients managing ADHD, abrupt discontinuation also produces a recognized withdrawal syndrome — fatigue, depressed mood, difficulty concentrating, sleep disruption — that the clinical literature classifies as stimulant discontinuation, distinct from addiction, but real and disruptive nonetheless.
The Telehealth Variable
One factor sustaining elevated demand deserves specific attention. The DEA’s pandemic-era waiver, which suspended the Ryan Haight Act’s requirement for an in-person evaluation before prescribing controlled substances, has been extended through the end of 2026.
That waiver was a reasonable public health accommodation during COVID-19. Its continued extension reflects both the political difficulty of rescinding it and the genuine access benefit it provides to patients in areas with limited psychiatric or primary care capacity. But it also means that the demand-side pressure that contributed to the original shortage has not been reduced. Every month the waiver remains in effect, the prescription volume the supply chain must serve continues at post-pandemic levels.
This is not an argument for ending the waiver — the patients it serves are real, and abrupt rescission would create its own access crisis. It is an observation that the demand-side and supply-side policy levers are not being managed in coordination. The DEA extended prescription access while simultaneously constraining production. The results have been predictable.
What Would Actually Fix It
The structural diagnosis has not changed since this series began. The DEA’s quota system is backward-looking by design — it calculates allowable production from historical demand data, in a market where demand has undergone a sustained structural shift. The system cannot respond in real time because it was not built to.
Legislative discussions on reforming quota-setting have not produced major legislation as of May 2026. What has changed is that the DEA’s willingness to make significant quota adjustments — the October 2025 increase was the largest in years — suggests the agency recognizes the system is undersupplying a legitimate medical need.
But recognition and resolution are not the same thing. The manufacturer exits since January represent a structural deterioration that a quota increase alone cannot reverse. Rebuilding generic manufacturing capacity for Schedule II substances requires investment decisions by private companies operating under regulatory constraints that have not materially changed. The companies that left the market did not leave because of the quota ceiling alone — they left because the economics of producing tightly regulated, low-margin generics under production caps that can shift administratively did not pencil out.
Until that equation changes — through quota reform, liability adjustments, API supply chain investment, or some combination — the shortage is not a problem being solved. It is a condition being managed. And right now, it is not being managed well.
Sources: Amphetamine mixed salts shortage data and manufacturer status from the American Society of Health-System Pharmacists Drug Shortage database (ashp.org/drug-shortages), updated continuously. DEA aggregate production quota figures from the agency’s annual quota orders published in the Federal Register, including the October 2025 order setting d-amphetamine at 26.5 million grams. FDA drug shortage notice for amphetamine mixed salts, originally posted October 2022, status current as of May 2026 (accessdata.fda.gov). Supply recovery timeline projections drawn from reporting by pharmacy trade and health policy press citing pharmaceutical supply chain analysts. CDC health advisories on counterfeit stimulant pill circulation issued 2024–2025 (cdc.gov). DEA telehealth waiver extension through December 2026 published via agency rulemaking notice. Ryan Haight Online Pharmacy Consumer Protection Act, 21 U.S.C. § 831.